Botox + Dysport
What is a BOTOX® Injection and What Does it Do?
BOTOX® injections utilize Botulinum Toxin Type A to treat wrinkles. BOTOX® was originally used to treat neurological disorders, but today it is also used for its cosmetic and aesthetic benefits.
BOTOX® Cosmetic is a simple, minimally-invasive, no-downtime procedure that relaxes dynamic wrinkles and can also treat excessive sweating conditions like hyperhidrosis.
How Does BOTOX® Work?
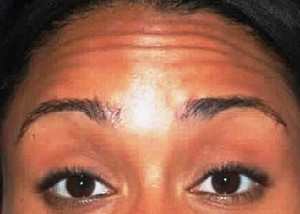
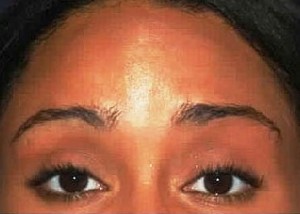
Botulinum Toxin Type A is a protein produced by the bacterium Clostridium botulinum. In medical settings, it is used as an injectable form of sterile, purified botulinum toxin. Small doses of the toxin are injected into the affected muscles and block the release of the chemical acetylcholine, which would normally signal the muscle to contract. Once the muscle is weakened and relaxed, it cannot contract. The lines gradually smooth out from disuse, and new creases are prevented from forming. The paralysis results in the improvement or disappearance of facial lines especially frown lines, “crow’s feet,” and forehead lines. Effects can be seen 3-5 days after treatment. BOTOX® is also effective in other areas such as the chin, neck, and décolletage (upper chest).
BOTOX® is also used to treat hyperhidrosis (excessive sweating). It works by temporarily blocking the chemical signals from the nerves that stimulate the sweat glands. When the sweat glands don’t receive chemical signals, the excessive sweating ceases.
The amount of Botox required to treat a given area is determined by the doctor or nurse providing the treatment. Typically, the forehead, glabellar area, and eyes can be treated with 40-45 units.
Is BOTOX® Safe?
Greater than 13 million aesthetic procedures have been administered with BOTOX® Cosmetic since the U.S. Food and Drug Administration approved its use in April of 2002 for temporary treatment of moderate to severe glabellar lines (the vertical “frown lines” between the eyebrows).
It is the only product of its type approved by the FDA.
Patients should always consult with their physician to see if treatment is right for them. Those with pre-existing neuromuscular disorders may be at increased risk of clinically significant systemic side effects from typical doses of BOTOX® Cosmetic.
What are the Side Effects of BOTOX® Injections?
There are very few potential side effects, and most side effects are temporary and typically associated with the injection itself, such as localized pain, tenderness, redness and/or bruising. In rare instances, patients may develop temporary weakness of the neighboring muscles or a headache.
How Long Will BOTOX® Last?
This depends entirely on the patient, but a single treatment of BOTOX® will normally be sustained for approximately 3-5 months. You will notice a gradual fading of its effect with time. Usually, BOTOX® treatment is required only three times a year.
Dysport™
Dysport™ is a wrinkle smoother similar to Botox®, and is used to treat moderate to severe frown lines. It is a form of Botulinum Toxin Type A, similar to the protein found in Botox® Cosmetic.
Dysport™ and Botox® are both cosmetic injections that temporarily relax the muscles in the face that create expressions, such as frown lines between the brows. These frown lines, if left untreated, can result in visible facial wrinkles and deeper creases.
Dysport™ is injected into the forehead and glabella (between the eyebrows) to relax muscles that create facial lines. It is a safe, entry-level procedure for those looking to create a more youthful look. Dysport™ and Botox® last approximately four months.
Contact us today to learn if Dysport™ or Botox® is right for you.

*There is no guarantee of specific results. Individual results may vary*

